 And the argument then goes that the reason for this is that if you add one or more extra electrons to the atom, inter-electron repulsions cause the atom to expand. It is perfectly true that negative ions have radii which are significantly bigger than the covalent radius of the atom in question. The problem comes in relating your choice of atomic radius to the "explanation" of the differences. You are, of course, perfectly free to compare the radius of an ion with whatever measure of atomic radius you choose. I want to focus on the non-metals, because that is where the main problem lies. The diagrams in the box above, and similar ones that you will find elsewhere, use the metallic radius as the measure of atomic radius for metals, and the covalent radius for non-metals. The 8-co-ordinated ionic radius for Cs is 0.174 nm rather than 0.167 for the 6-co-ordinated version. CsCl actually crystallises in an 8:8-co-ordinated structure – so you couldn't accurately use these values for CsCl. Just use the values you are given in whatever units you are given.įor comparison purposes, all the values relate to 6-co-ordinated ions (the same arrangement as in NaCl, for example). A value in pm will look like, for example, for chlorine, 181 pm rather than 0.181 nm. You may also come across tables listing values in pm (picometres) which are 10 -12 m. To convert from Angstroms to nm, you have to divide by 10, so that 1.02 Angstroms becomes 0.102 nm. If you are interested, 1 Angstrom is 10 -10 m 1 nm = 10 -9 m. I have converted them from Angstroms to nm (nanometres), which are more often used in the data tables that you are likely to come across. 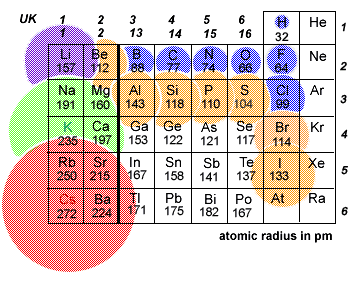
Note: These figures all come from the Database of Ionic Radii from Imperial College London. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
